 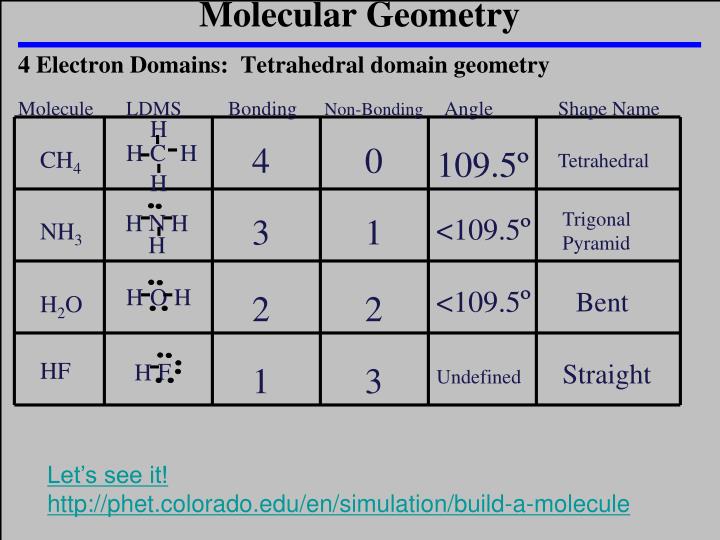
Determine the electron domain geometry, molecular geometry, and bond angles. Count the electron domains, and determine whether they are bonding or non-bonding pairs. PRACTICE PROBLEMS : Use the electron dot structure (Lewis structure) and the electron geometry table to determine the steric number and electron geometry. To predict the molecular geometry, follow these steps: 1. What is the steric number and electron geometry of PH5? What is the steric number and electron geometry of BrF3? What is the steric number and electron geometry of SH2? 
What is the steric number and electron geometry of SO2? The bond angles are affected by all electron domains, but they only describe the angle between bonding electrons. Lone pair of electrons can change the bond angles due to their repulsive forces, but here in C2H6, as there are no lone pairs in the molecule, the bond angles in C2H6 is 109.5 degrees. Bond angles (BA) - The angle between two adjacent bonds in the same atom. The molecules with a tetrahedral molecular geometry have bond angles of 109.5 degrees, which are typically affected by lone pairs of electrons. Refer to the electron geometry table below.Įxamples: Use the electron geometry table to determine the steric number and electron geometry of each molecule. Molecular geometry - The arrangement of the atoms in a molecule (The nonbonding domains are not included in the description). Therefore, according to the VSEPR chart or AXE notation, the electron geometry for NH 3 is a Tetrahedral and its molecular geometry is Trigonal pyramidal. This means you count up the lone pairs and number of atoms attached to the central atom into one number (the steric number) and from there determine electron geometry. As per the VSEPR chart, if a molecule gets AX 3 N 1 generic formula then its molecular geometry or shape will be a trigonal pyramid, and its electron geometry will be tetrahedral. You also treat double and triple bonds as one group instead of 3 just like you did in molecular shape. However, with electron geometry (steric number) lone pairs and bonds are treated the same. If we look at the nitrite ion NO2-, we have 2 Bond Pairs and 1 Lone pair of electrons. In NO2, we have 2 Bond Pairs and 1 lone electron. In molecular shape (molecular geometry) you treat the electron’s lone pairs and the bonds as two different and separate things. VSEPR chart: We can see that NO2 has a bent molecular geometry and the angle is around 120 degrees. What is the difference between electron geometry (electron domains / electron clouds / steric number) and molecular shape (molecular geometry)?īoth are very similar, but the difference is in how we treat that electron’s lone pairs versus the bonds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
